Desde la protección de los ingredientes activos hasta la mejora de la gestión de los medicamentos por parte del paciente, el dióxido de titanio (TiO2) desempeña un papel esencial en todas las etapas del ciclo de vida de un producto farmacéutico. Si bien el TiO2 ha sido durante mucho tiempo un ingrediente clave en innumerables bienes de consumo e industriales, el papel que ocupa en la industria farmacéutica es posiblemente el más vital. Gracias a sus propiedades altamente deseables, el TiO2 se utiliza en la gran mayoría de medicamentos del mercado. Según la Agencia Europea de Medicamentos (EMA), no existe un solo material que proporcione la misma combinación de propiedades que son exclusivas del TiO2

Las propiedades beneficiosas únicas del TiO2 en medicamentos
El TiO2 no solo se utiliza con fines estéticos. Realiza una combinación única de funciones clave en todas las etapas del ciclo de vida de un producto farmacéutico. A lo largo de un siglo, esto lo ha hecho indispensable para fabricantes, médicos y pacientes de todo el mundo, y hoy en día el TiO2 está casi omnipresente en medicamentos esenciales para humanos.
Algunas de las propiedades y aplicaciones clave del TiO2 incluyen:
- Recubrimientos protectores para preservar la eficacia de los medicamentos a lo largo del tiempo – El TiO2 es un componente esencial de los recubrimientos protectores de los medicamentos, que mejora la seguridad, eficacia y calidad de los productos farmacéuticos durante periodos más prolongados. Gracias a su capacidad para dispersar la luz y absorber los rayos UV, el TiO2 prolonga la vida útil y garantiza la estabilidad de los productos farmacéuticos al proteger los principios activos contra la degradación por los rayos UV/luz y el calor.
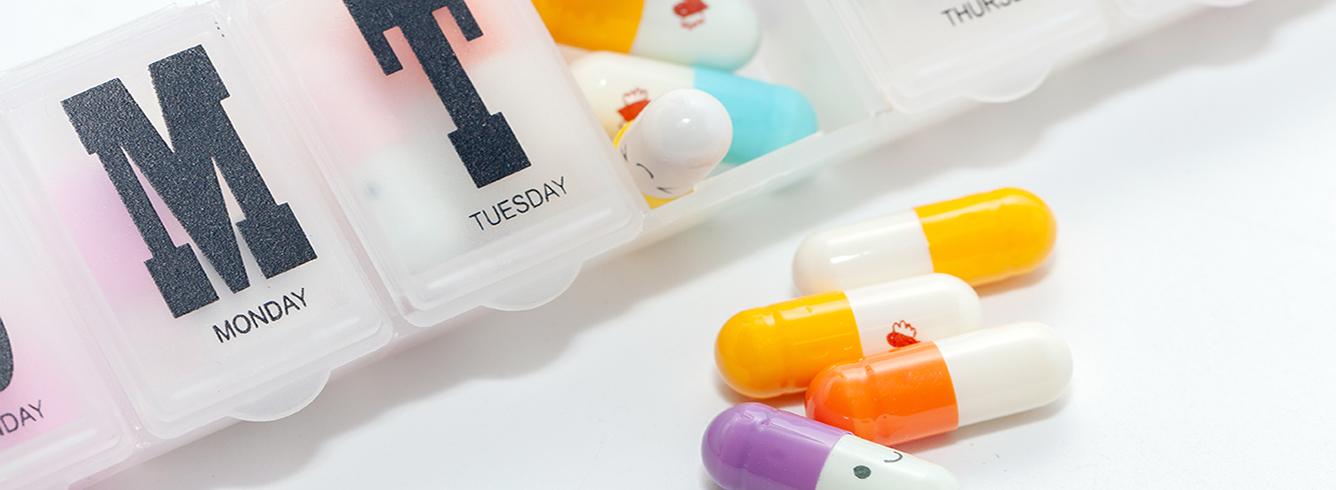
- El TiO2 hace que los productos farmacéuticos sean más seguros de usar y más fáciles de gestionar: tanto si se utiliza para añadir blancura como para acentuar la audacia de otros colores en los recubrimientos protectores utilizados en los medicamentos, el TiO2 permite una amplia gama de colores y garantiza que los medicamentos mantengan la uniformidad del color a lo largo del tiempo. La consistencia y la uniformidad del color desempeñan un papel importante en la capacidad de los pacientes y los profesionales médicos para diferenciar los medicamentos y las dosis y gestionar con seguridad sus medicamentos. Esto tiene un impacto positivo directo en la seguridad del paciente y en el cumplimiento de las instrucciones de prescripción. Esto es crucial para las personas con visión limitada, o para aquellas que necesitan varios medicamentos diferentes o cuando existen diferentes concentraciones del mismo medicamento, como es el caso del anticoagulante común warfarina, en el que cuatro colores distintos denotan diferentes dosis. La coloración también es una indicación clave para los profesionales sanitarios y los centros de urgencias a la hora de responder a casos de sobredosis o intoxicación por medicamentos, y puede ayudar a determinar si un comprimido es auténtico o falsificado.
- Ingrediente inerte y eficaz – El TiO2 es una sustancia inerte y no reactiva que no interfiere con las propiedades de los principios activos de los medicamentos ni con otros componentes esenciales no activos de un medicamento (excipientes). Sólo se necesita una pequeña cantidad de TiO2 para conseguir las propiedades deseadas. Habría que añadir materiales alternativos en mayores cantidades, sin que por ello desempeñaran las mismas funciones que el TiO2